Where to Find CAR-T Cell Therapy for Blood Cancers: A Global Overview
CAR-T cell therapy and gene therapy are revolutionary medical breakthroughs that have proved effective in treating previously untreatable hematological malignancies. The success story of CAR-T therapy began with a profound idea in 1989, culminating in the miraculous recovery of 6-year-old Emily Whitehead in 2012, who was considered a lost cause at the time. Emily is still alive today, in 2023.
While there is no one "best" country for CAR-T cell therapy, the availability of the treatment in a patient's home country, as well as their individual needs and circumstances, are crucial factors to consider. Consultation with the healthcare professional treating the patient in their home country is essential to determine the best course of treatment for each patient.
What countries offer FDA Approved Car-T therapy?
Several countries, including the USA, Canada, Germany, France, Spain, the United Kingdom, Italy, ISRAEL, Australia, Singapore, and China, offer FDA-approved CAR-T therapy. However, there are some obstacles to receiving the treatment, such as long waiting times for citizens of the country and high costs, starting at approximately $475,000, due to the need for the patient's blood to travel to the United States under strict freeze conditions for gene engineering before returning to the patient.
Israeli hospitals offer excellent CAR-T programs, leveraging world-renowned Israeli medicine, achieving success rates comparable to those reported in the literature, and providing the most cost-effective options.
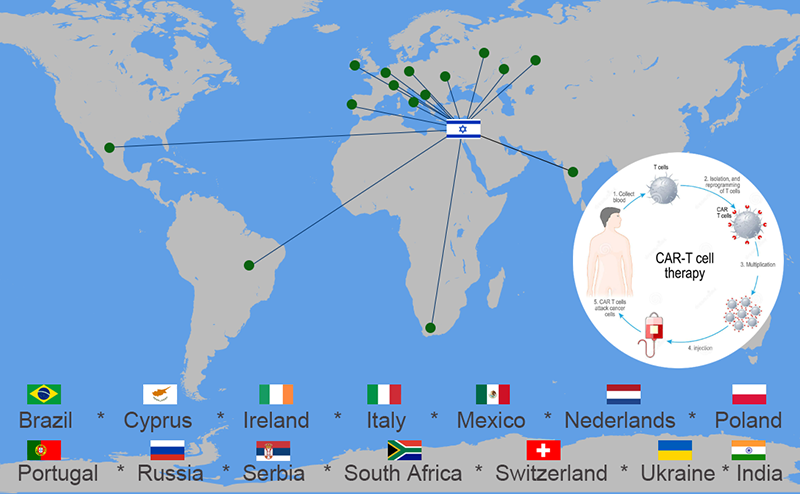
CAR-T programs offered as a part of clinical trials
To make CAR-T therapy available to more patients, some medical centers in the USA, China, and Israel offer in-house CAR-T therapy at significantly lower price ranges. In-house CAR-T, or home-grown CAR-T, is created in the medical center's laboratories, preserving the quality of the cells and expediting the procedure.
China and the USA lead in the number of clinical studies involving CAR-T, targeting hematological antigens CD19, BCMA, and more. As of March 10, 2022, China has the highest number of CAR-T cell clinical trials registered on ClinicalTrials.gov, followed by the US, the UK, and Germany, with 25 countries initiating clinical trials with CAR-T cells. Non-Hodgkin's lymphoma is still the most frequent hematological disease targeted by CAR-T cell therapy, followed by B-ALL, Multiple Myeloma, chronic lymphocytic leukemia, and acute myeloid leukemia, while T-cell cancers and Hodgkin's disease are still the minority.
Sources:
NIH- National Library of Medicine
Single Use Support- biopharma
WHAT ARE THE KEY FACTORS TO CONSIDER
WHEN CHOOSING A CAR-T THERAPY PROVIDER FOR INTERNATIONAL PATIENTS?
Testimonial by a CAR-T patient at Hadassah hospital

My Husband and I engaged with Tanya Preminger from IHA to travel from New Zealand to Israel for undertaking Car T treatment. During the preparation stage Tanya was very professional in her approach and extremely responsive to our many emails. During our 8 week stay in Israel Tanya was incredibly supportive on both a practical and social level. Tanya was an effective communicator with ourselves and the Hadassah hospital staff, always accountable and flexible with our needs. We would highly recommend Tanya as a representative for other clients who may be considering various treatments in Israel.
Yvonne and Mike Hogan, New Zealand, 8/5/2023
One more patient completed CAR-T cell therapy for blood cancer ( June 2024).

With Dr. Marcus at Sheba hospital, celebrating cancer-free PET CT. (2024)

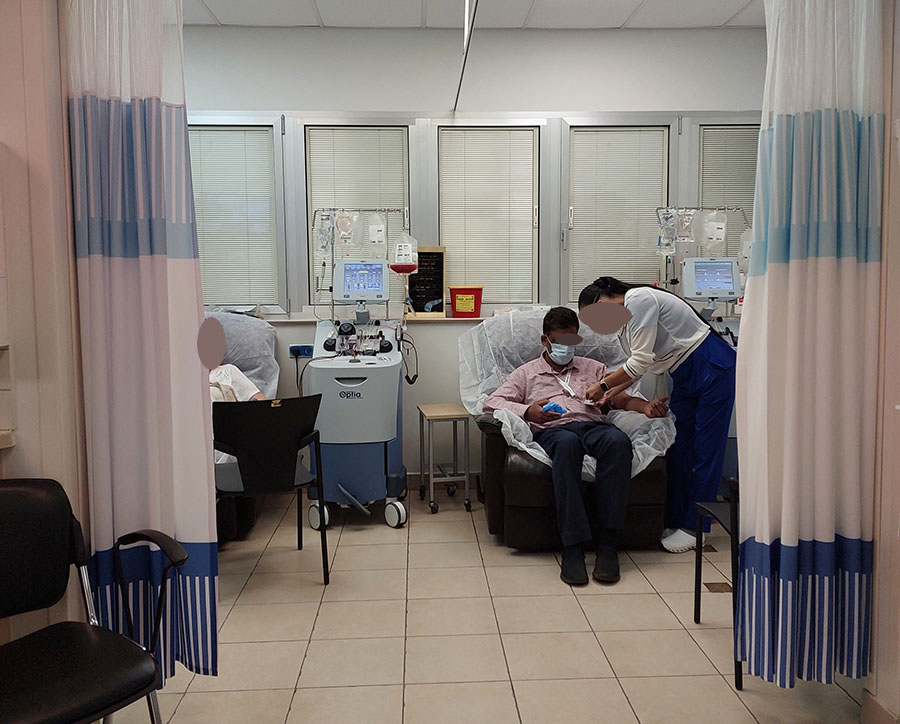
A patient during CAR-T therapy in Sheba hospital, Israel
Testimonial by Lymphoma patient who achieved complete metabolic response after CAR-T
We had a great and seamless experience working with Tanya. For us the idea of going through a treatment in foreign land was daunting but Tanya made everything feel like home and comfortable. She went above and beyond to help us at each step. She helped with finding a right place to stay, with interactions with medical staff and also with any adhoc request that was not part of the original services. She is a great and kind person to work with and we are grateful for all her help.
October 2023
Links
CAR-T cell therapy in Israel
CAR-T for Leukemia and Lymphoma in Israel
CAR-T for Myeloma in Israel
CAR-T cell therapy for leukemia and non-Hodgkin's lymphoma (NHL) in Europe
CAR-T cell therapy for Multiple Myeloma patients in Europe
CAR-T therapy for international patients
CAR-T therapy in China
CAR-T therapy in Spain
CAR-T cell therapy in Germany
CAR-T cell therapy in Japan

CAR-T testimonials >>
Our team travelled to Strasbourg, France, to attend #CART25 – one of the most important conferences in European hematology. This event brought together the world's leading specialists to discuss the latest advancements in CAR-T therapy, a groundbreaking treatment transforming hematology and cancer care.










